Calorie Restriction: A Double-Edged Sword for Fat Cells and Weight Loss
- S A

- May 5, 2024
- 6 min read
Updated: Aug 4, 2025
Calories: Part 2
Shedding pounds often involves cutting calories, but new research suggests this approach might be a double-edged sword. While it can lead to weight loss initially, it might also trigger hormonal changes that make it harder to keep the weight off in the long run. This blog post dives into a study published in the National Institutes of Health's National Center for Biotechnology Information: [1], exploring the fascinating interplay between fat cells, hormones, and calorie restriction.
From Food to Fat: The Body's Storage Strategy
The food we eat is broken down into components our body can use for energy or store for later. Excess calories, particularly carbohydrates and fats, are converted into triglycerides, a type of fat, and shuttled to fat cells for storage.
Lipoprotein Lipase: The Fat Cell Gatekeeper
Lipoprotein lipase (LPL) acts as a security guard for fat cells. This enzyme functions like a key, unlocking the door to allow fat (triglycerides) circulating in the bloodstream to enter the fat cells for storage.
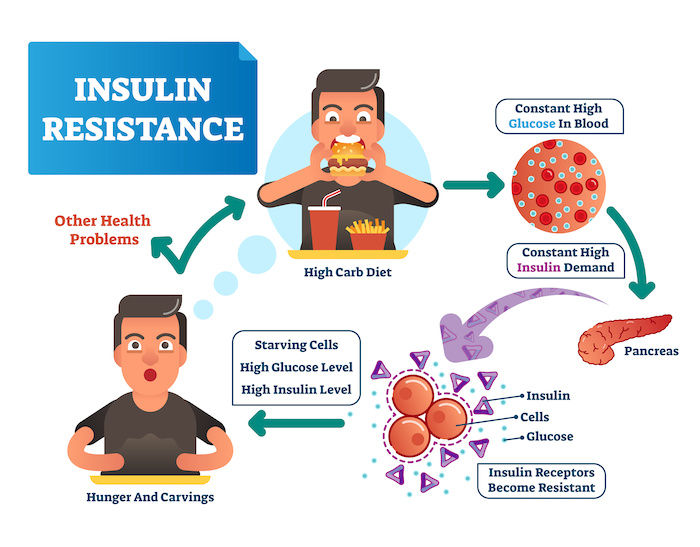
Insulin: The Master Regulator of Fat Storage
The hormone insulin plays a key role in fat storage. When insulin levels are high, it acts as a messenger, sending a signal to fat cells to open their doors via LPL activity and take up fat from the bloodstream. Conversely, low insulin levels signal fat cells to shut their doors and restrict fat uptake.

Credit: Gene-Medium
Why Cutting Calories Might Not Be the Whole Story: The Hungrier Fat Cell
The National Institutes of Health study investigated the effects of long-term calorie restriction on fat cells. They found that:
Reduced LPL Activity During Weight Loss: When overweight women were put on a calorie-restricted diet for three months, their fat cells showed a significant decrease in LPL activity. Prior to calorie restriction, LPL activity was measured at 2.0 nmol/g/h. After three months of calorie restriction, this number dropped to 0.7 nmol/g/h, a decrease of almost two-thirds. This indicates that the fat cells were less eager to take up fat from the bloodstream during weight loss.
The Rebound: Fat Cells Fight Back
Here's where things get interesting. When the women transitioned to a maintenance diet to sustain their weight loss, the researchers observed a significant increase in LPL activity. In essence, the fat cells became hungrier, potentially due to the body adapting to the lower calorie intake. LPL activity levels jumped to a staggering 7.8 nmol/g/h, a nearly 12-fold increase compared to the post-calorie restriction levels. This can make it challenging to maintain weight loss because the body is working harder to store fat.
Beyond Calories: Insulin as a Potential Key Player
Rather than obsessing over calories, focusing on lowering insulin levels might be a more effective strategy for weight management. Here's why:
Low Insulin, Less Fat Storage: When insulin levels are low, fat cells become less receptive to storing fat. At the cellular level, insulin acts like a docking station for fatty acids (the building blocks of fat) to enter the fat cell. With low insulin, there are fewer docking stations available, and fatty acids are less likely to be ushered into the cell for storage. Insulin stimulates the production of lipoprotein lipase (LPL) on the surface of fat cells. LPL breaks down triglycerides (circulating fat) into fatty acids, which can then enter the fat cell for storage. When insulin levels are low, less LPL is produced, hindering the breakdown and storage of fat.

Credit: Healingfatsoflife
The Downstream Effects: A Hormonal Response to Calorie Restriction
Cutting calories can trigger a cascade of hormonal changes that work against weight loss efforts:
Ghrelin on the Rise: Ghrelin, often referred to as the "hunger hormone," increases when calories are restricted. This can lead to increased feelings of hunger, making it harder to stick to a reduced-calorie diet.
Satiety Hormones Take a Hit: Calorie restriction can also decrease the production of satiety hormones like leptin, which signal feelings of fullness. The brain constantly monitors hormone levels, and leptin plays a crucial role in regulating appetite. When leptin levels are low, the brain interprets this as a sign of hunger, triggering cravings and making it harder to feel satisfied after eating. This creates a double whammy: increased hunger due to ghrelin and decreased feelings of fullness due to low leptin, making it more challenging to maintain a calorie deficit.
LPL Up, Metabolism Down: As discussed earlier, calorie restriction can lead to increased LPL activity, promoting fat storage. Additionally, the body might adapt by lowering metabolism in response to restricted calories, further hindering weight loss efforts.

Credit:Cell Metabolism
Fat Cells: More Than Just Storage Units
The study highlights the remarkable adaptability of fat cells. They are not simply passive storage compartments but dynamic players that respond to hormonal signals and calorie intake. Understanding this cellular communication is crucial for developing effective weight management strategies.
The Unsaved Fat's Destination: Muscle to the Rescue
If fat cells become resistant to taking up fat due to low insulin levels, where does the excess fat from food go?
Muscle Takes Up the Slack: When insulin is low and fat cells are less receptive, muscle cells can step up and take in some of the circulating fat. Muscle tissue has the ability to burn fat for energy, potentially aiding in weight loss. However, it's important to note that muscle cells have a limited capacity for fat storage compared to fat cells.
The Metabolic Rate Maze: The Impact of Calorie Restriction
Calorie restriction can lead to a decrease in metabolic rate. This means the body burns fewer calories at rest, making it harder to maintain weight loss. This is another reason why focusing on strategies that address hormonal imbalances, like insulin levels, might be more effective for long-term weight management.
Calorie Restriction and Lowered Metabolism
Survival Mode: When calories are consistently restricted, the body perceives it as a starvation state. To conserve energy and protect vital organs, the body makes adjustments to its metabolic rate, burning fewer calories at rest. This is a survival mechanism to ensure enough energy remains for critical functions.
Thyroid Hormone Regulation: The thyroid gland plays a key role in regulating metabolism. Calorie restriction can suppress the production of thyroid hormones, further slowing down the metabolic rate.
Muscle Loss: Muscle tissue is metabolically active, meaning it burns calories even at rest. During calorie restriction, the body might break down muscle for energy, leading to a decrease in overall metabolic rate. Interestingly, research suggests that muscle loss might be less pronounced during fasting compared to calorie restriction. Here's why: An Evolutionary Perspective From an evolutionary standpoint, it makes sense that muscle wouldn't be the primary target during periods of food shortage. Our ancestors relied on muscle strength for hunting, gathering, and survival. Losing muscle mass during times of limited food intake would have been detrimental, making it difficult to find sustenance. The body's adaptations to preserve muscle during fasting might be a legacy of our evolutionary past.
Growth Hormone Boost: Fasting can lead to a significant increase in human growth hormone (HGH) production. HGH is an anabolic hormone that helps build and preserve muscle mass. A review paper published in the journal Nutrients in 2021 summarized findings from various studies. The review highlights that in humans, fasting for 37.5 hours elevates basal GH concentrations by 10-fold and reduces the metabolic clearance rate of GH, essentially making HGH more available . This increase in HGH during fasting may help counteract muscle breakdown. [2].
Protein Recycling: The body has a remarkable ability to adapt during periods of food scarcity. It can recycle protein from non-essential tissues to provide amino acids for critical functions, minimizing muscle breakdown.
Calorie Abundance and Increased Metabolism
Energy Availability: When there's an abundance of calories, the body doesn't need to go into calorie-saving mode. It can readily access energy from food, leading to a higher metabolic rate to process and utilize the incoming fuel.
Thermogenic Effect of Food: Digesting and absorbing food requires energy itself. This energy expenditure, known as the thermic effect of food (TEF), contributes to a slightly increased metabolic rate after eating. However, the thermic effect is relatively small compared to the overall metabolic rate.
Rethinking Weight Loss Strategies
The interplay between fat cells, hormones, and calorie intake is complex. While calorie restriction can lead to initial weight loss, it might trigger adaptations that make it difficult to keep the weight off. Focusing on dietary approaches that promote healthy insulin levels and hormonal balance, along with regular exercise to build muscle mass, might be a more sustainable strategy for achieving and maintaining a healthy weight.
[1] Weight reduction increases adipose tissue lipoprotein lipase responsiveness in obese women. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC442337/
[2] Effects of Intermittent Fasting on the Circulating Levels and Circadian Rhythms of Hormones. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8419605/





Comments